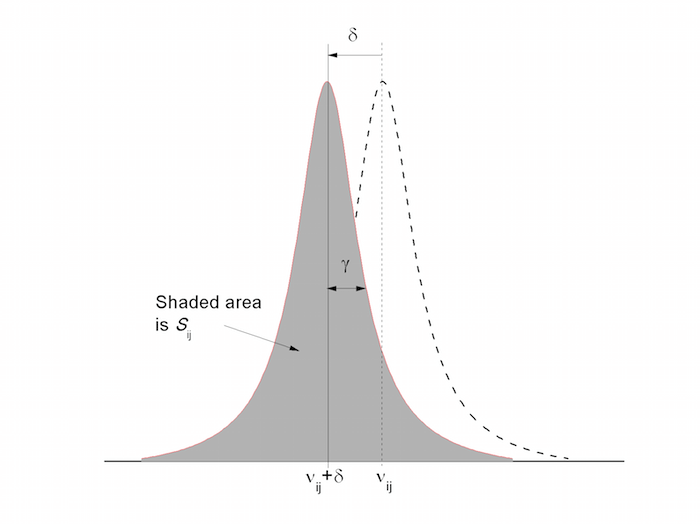
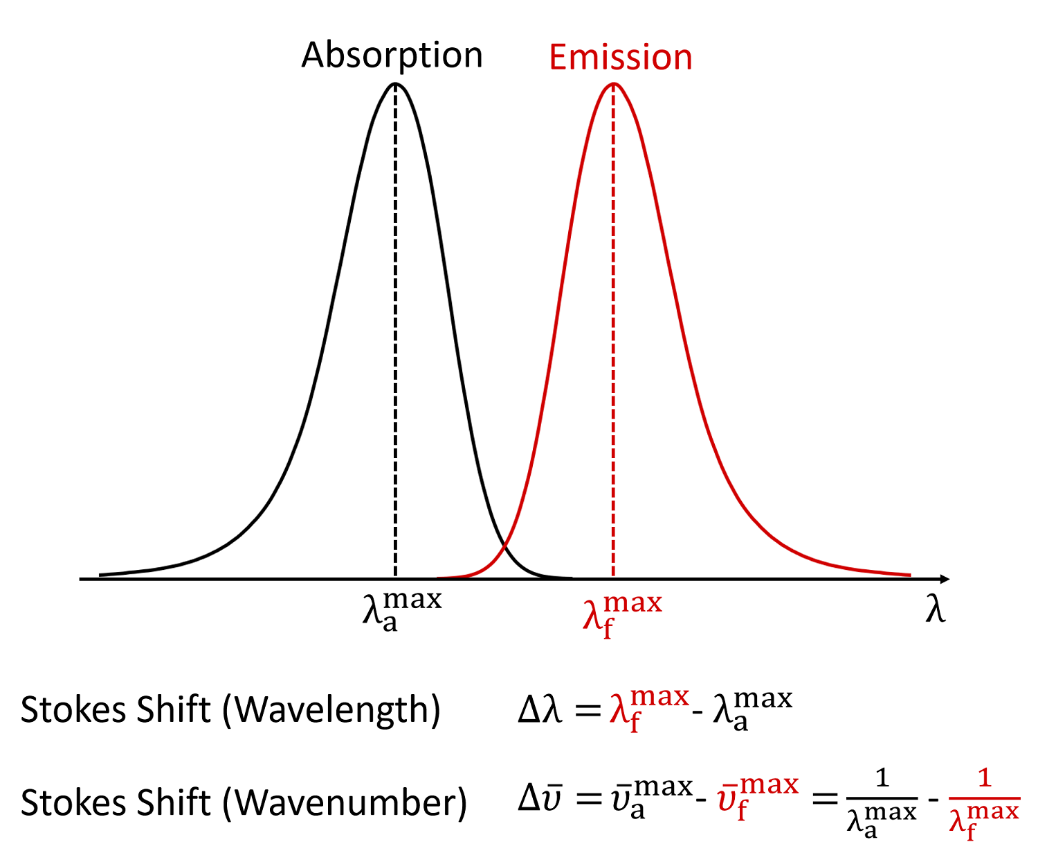
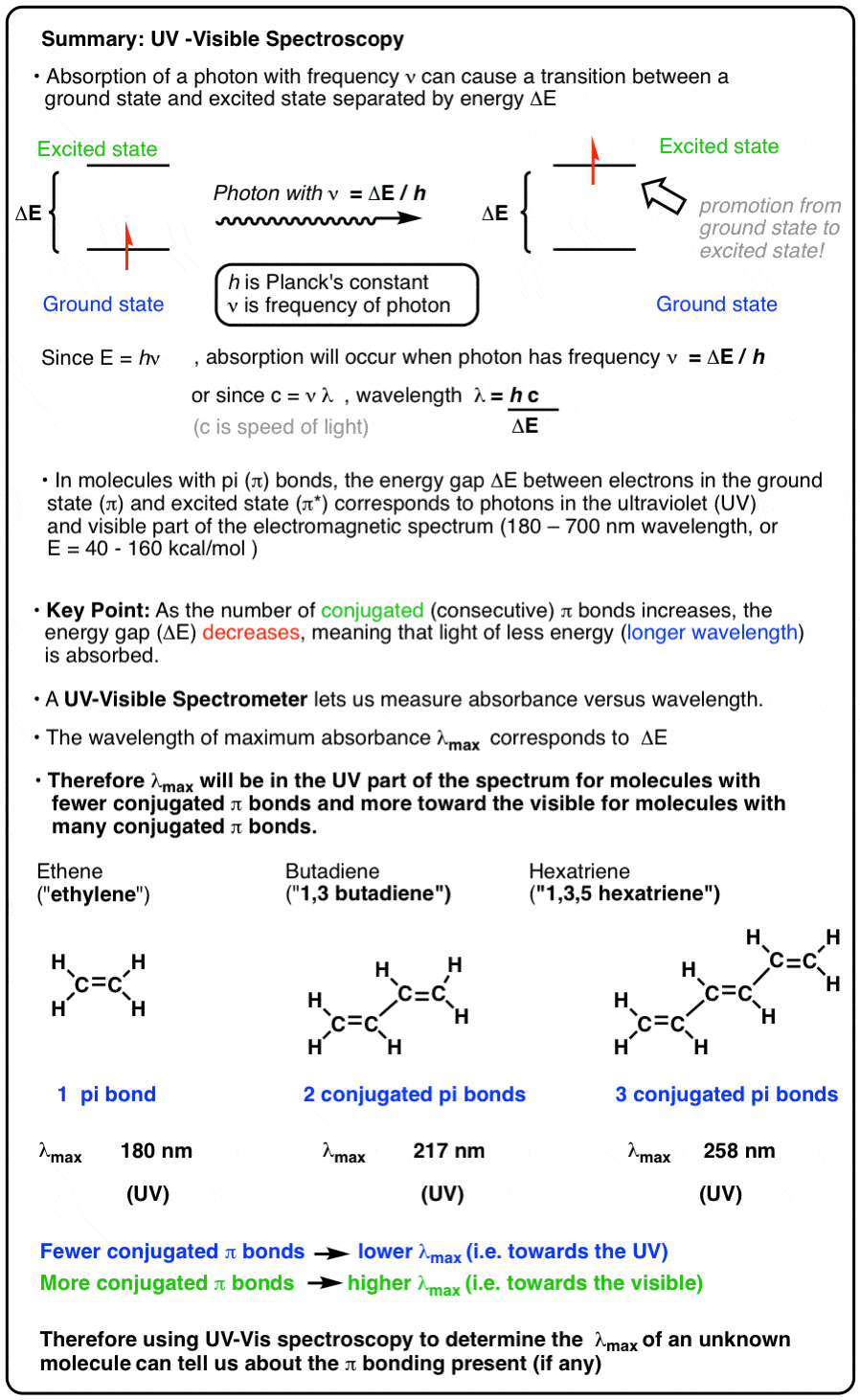
CALCULATION OF THE TRANSITION DIPOLE MOMENT OF THE $\tilde{A}\leftarrow \tilde{X}$ ELECTRONIC TRANSITION OF THE C$_2$H$_5$O$_2$ FROM THE PEAK ABSORPTION CROSS-SECTION
CALCULATION OF THE TRANSITION DIPOLE MOMENT OF THE $\tilde{A}\leftarrow \tilde{X}$ ELECTRONIC TRANSITION OF THE C$_2$H$_5$O$_2$ FROM THE PEAK ABSORPTION CROSS-SECTION

The longest wavelength doublet absorption transition is observed at 589 and 589.6 nm. Calculate the frequency of each transition and energy difference between two excited states.

CHEM1011 notes for 8 topics - Atomic spectra & Rydberg 1. Relate the lines and series in the - Studocu
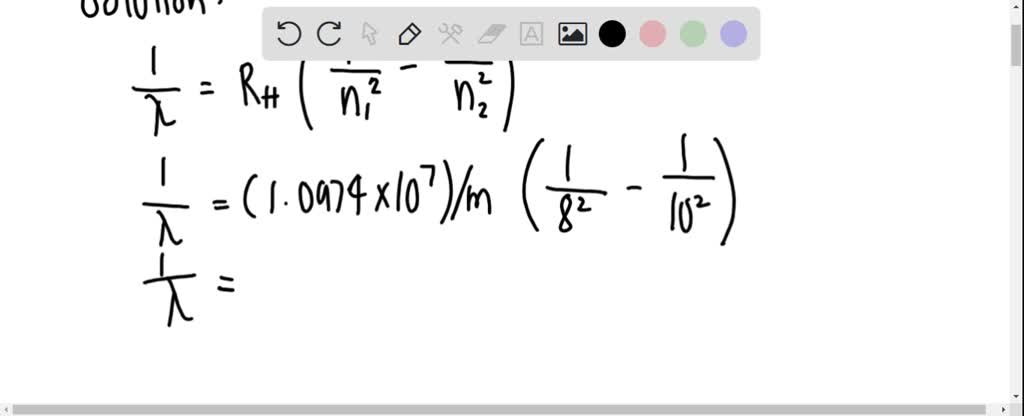
SOLVED: Calculate the wavelength of the line in the absorption line spectrum of hydrogen caused by the transition of the electron from an orbital with n=8 to an orbital with n=10. Round

SOLVED: 2) Which of these transitions correspond to emission and which to absorption? a. n = 2 to n = 4, Absorption b n = 3to n = 1, Emission C. n =

Calculate the wavelength of the line in the absorption line spectrum of hydrogen caused by the transition of the

In a one-body picture, there is only one transition in the absorption... | Download Scientific Diagram
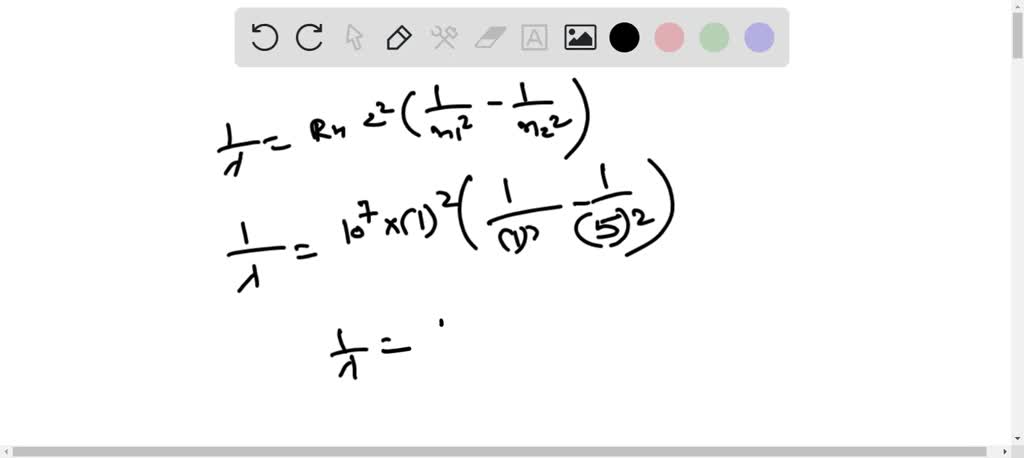
SOLVED: Calculate the wavelength of the line in the absorption line spectrum of hydrogen caused by the transition of the electron from an orbital with n=1 to an orbital with n=5. Round

First-principles modelling of the L-edge X-Ray Absorption Spectroscopy of Transition Metal Oxides and Organic Molecules with Transition Metals Centers – eSSENCE
CALCULATION OF THE TRANSITION DIPOLE MOMENT OF THE $\tilde{A}\leftarrow \tilde{X}$ ELECTRONIC TRANSITION OF THE C$_2$H$_5$O$_2$ FROM THE PEAK ABSORPTION CROSS-SECTION
![PDF] Calculation of single-beam two-photon absorption transition rate of rare-earth ions using effective operator and diagrammatic representation | Semantic Scholar PDF] Calculation of single-beam two-photon absorption transition rate of rare-earth ions using effective operator and diagrammatic representation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b0045c9a6c975d5467e09bdc57a57de9752d40b5/8-Figure1-1.png)