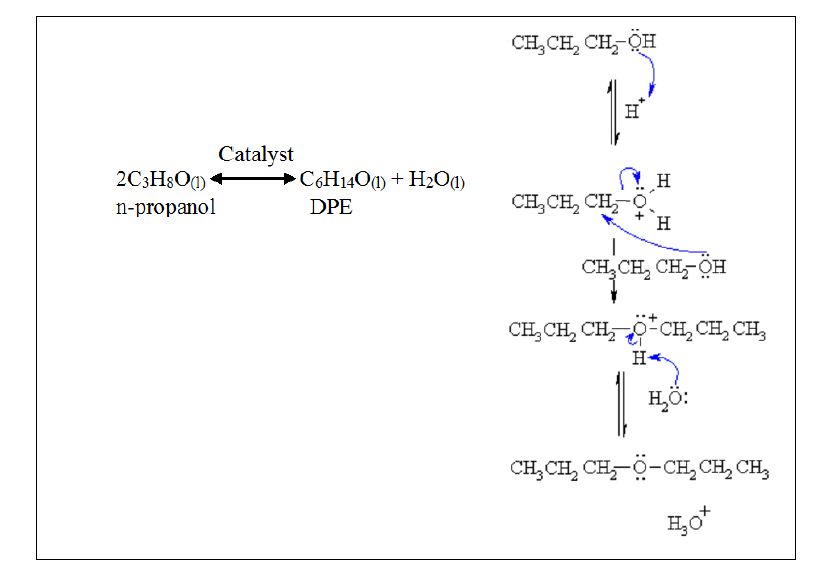
What unsaturated carbonyl compound is formed by dehydration of the below beta-hydroxy carbonyl compound? | Homework.Study.com
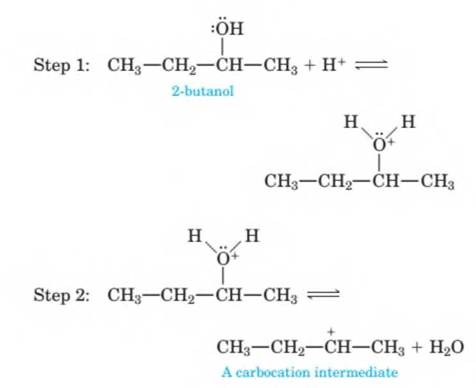
14-71 The mechanism of the acid-catalyzed dehydration of an alcohol to an alkene is the reverse of the acid- catalyzed hydration of an alkene. The dehydration mechanism occurs by the following three

PDF) Efficient Catalytic Dehydration of High-Concentration 1-Butanol with Zn-Mn-Co Modified γ-Al2O3 in Jet Fuel Production
![PDF] Heat Capacities and Derived Thermodynamic Functions of 1-Hexanol, 1-Heptanol, 1-Octanol, and 1-Decanol between 5 K and 390 K | Semantic Scholar PDF] Heat Capacities and Derived Thermodynamic Functions of 1-Hexanol, 1-Heptanol, 1-Octanol, and 1-Decanol between 5 K and 390 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a837643494d3816eb68e684fd65ecde2133919c5/6-Table16-1.png)
PDF] Heat Capacities and Derived Thermodynamic Functions of 1-Hexanol, 1-Heptanol, 1-Octanol, and 1-Decanol between 5 K and 390 K | Semantic Scholar

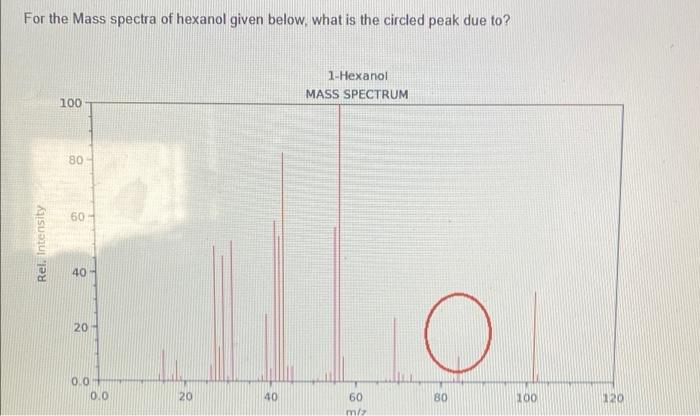
SOLVED: Draw the reactants and products for each of the chemical rcactions below: Dehydration of pure hexanol at 180*€ in the presence of H,SOs Dehydration of a mixture ethanol and [-propanol at

Saline-Sodium Citrate (SSC), 20X Solution (Molecular Biology), Fisher BioReagents Nalgene Poly Bottle; 1L Saline-Sodium Citrate (SSC), 20X Solution (Molecular Biology), Fisher BioReagents | Fisher Scientific

Compound X is 2-propanol. When compound X is heated with a strong acid, it dehydrates to compound Y (C_3H_6). When compound X is oxidized, compound Z is forms which cannot be oxidized

SOLVED: Spccify both the alcohol starting ' matcrial and the reagents you would use in cach stcp in a synthesis of the compound shown: If the synthesis requires only two steps enter
S 1 Reaction Remember that a 3˚ alkyl halide will not undergo a S 2 reaction The steric hindrance in the transition state

Separation and Purification of Methyl Isobutyl Ketone from Acetone + Isopropanol + Water + Methyl Isobutyl Ketone + Methyl Isobutyl Carbinol + Diisobutyl Ketone Mixture | ACS Omega
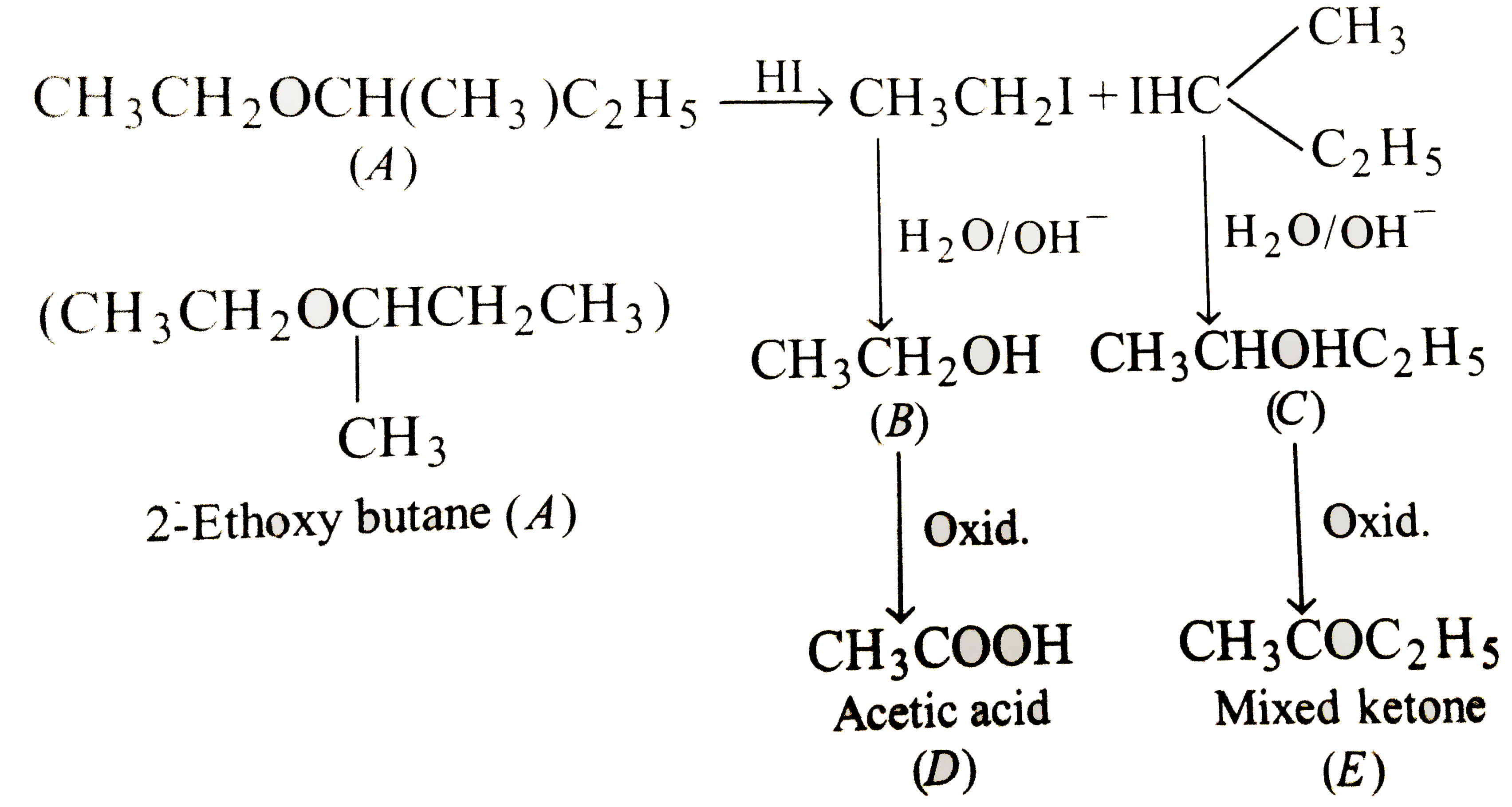
An ether, (A) having molecular formula, C(6)H(14)O, when treated with excess of HI produced two alkyl iodides which on hydrolysis yield compounds (B) and (C). Oxidation of (B) gives an acid (D),



![PDF] Combinatorics of symmetric plabic graphs | Semantic Scholar PDF] Combinatorics of symmetric plabic graphs | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/39f2f4dfdfdabbae5b432f4b71f2fd20dce03998/8-Figure4-1.png)